Dupilumab Tied to Facial, Neck Erythema
By Dermsquared Editorial Team | April 28, 2021
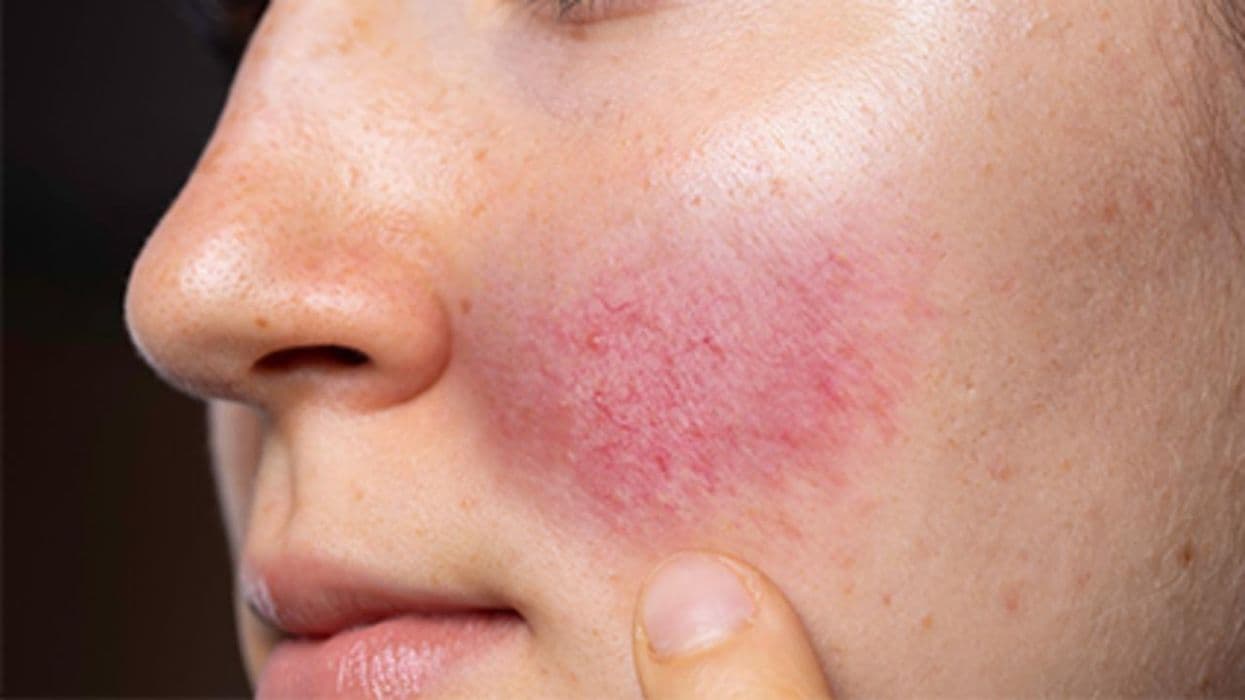
Some patients taking dupilumab develop facial or neck erythema that differs from their usual atopic dermatitis symptoms, according to a review published in the May issue of the Journal of the American Academy of Dermatology.
Christine E. Jo, from the University of Ottawa in Toronto, and colleagues sought to characterize cases of reported dupilumab-associated facial or neck erythema, with a focus on etiology and management strategies.
The researchers identified 101 patients from 16 studies who had dupilumab-associated facial or neck erythema. Half (52 percent) had baseline atopic dermatitis facial or neck involvement, and 45 percent reported different cutaneous symptoms from preexisting atopic dermatitis, possibly suggesting a different etiology, including rosacea, allergic contact dermatitis, and head and neck dermatitis. Treatments most commonly included topical corticosteroids, topical calcineurin inhibitors, and antifungal agents. Of the 57 patients with data on the course of adverse events, 29 had improvement, four had clearance, 16 had no response, and eight reported worsening. This facial or neck erythema caused 11 percent of the patients to discontinue dupilumab.
"Dupilumab-associated facial or neck erythema should be considered in patients presenting with facial symptoms atypical of baseline disease, especially after original clearance or divergence from the improvement observed in other regions," the authors write. "Educating patients about this adverse event before initiation may allow prompt identification and early management, which may minimize distress or discontinuations in patients who are otherwise satisfied with their dupilumab treatment."
Several authors disclosed financial ties to the pharmaceutical industry.
Abstract/Full Text (subscription or payment may be required)