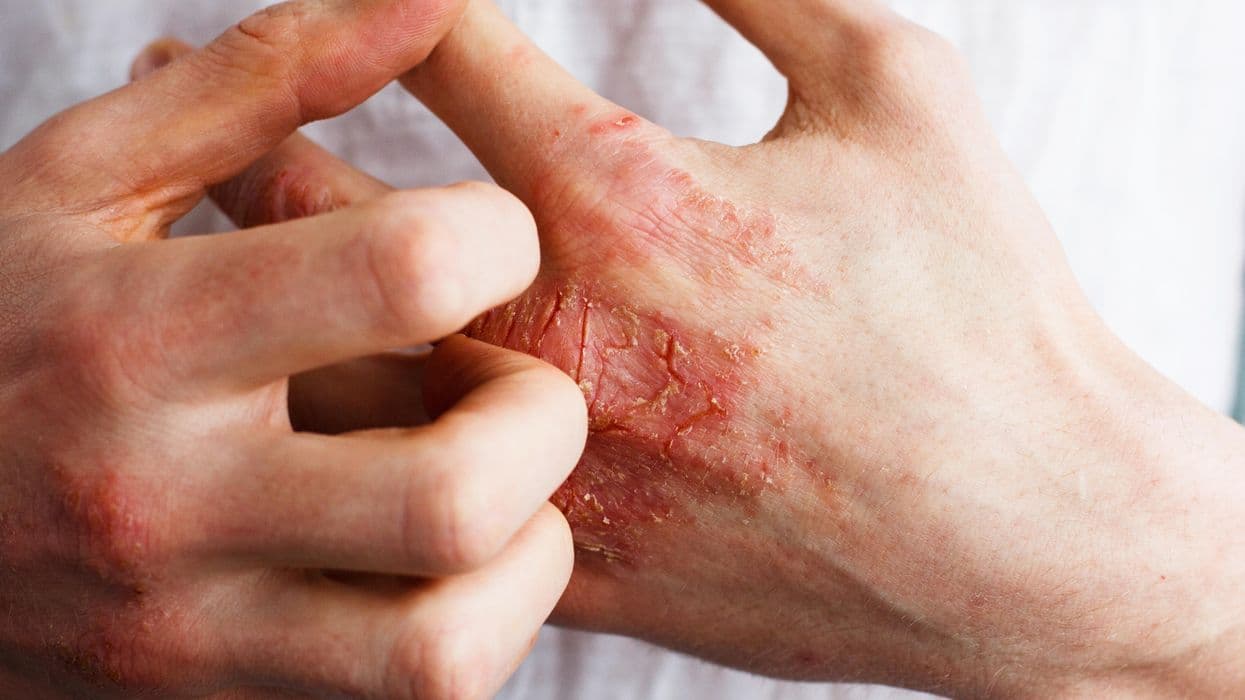
Is VTE Risk Increased With Atopic Dermatitis, JAK Inhibitor Use?
By Dermsquared Editorial Team | August 24, 2022
Patients with atopic dermatitis (AD) do not seem to have an increased risk for venous thromboembolism (VTE), nor is treatment with Janus kinase (JAK) inhibitors associated with an increased risk for VTE in AD, according to a review and meta-analysis published online Aug. 24 in JAMA Dermatology .
Tai-Li Chen, M.D., from Taipei Veterans General Hospital in Taiwan, and colleagues examined the association of AD with incident VTE and assessed the risk for incident VTE among AD patients receiving treatment with JAK inhibitors in a systematic review. Data were included from two cohort studies and 15 randomized controlled trials with 466,993 participants.
The researchers found that in a meta-analysis, there was no significant association between AD and incident VTE (hazard ratio, 0.95; 95 percent confidence interval, 0.62 to 1.45). Overall, 0.05 percent of the 5,722 patients with AD who were receiving treatment with JAK inhibitors experienced VTE compared with 0.03 percent of the 3,065 patients with AD receiving placebo or dupilumab (Mantel-Haenszel risk difference, 0). The incidence rate of VTE among participants with AD receiving JAK inhibitors and placebo was 0.15 and 0.12 events per 100 patient-years, respectively. Similar findings were seen for four unique JAK inhibitors (abrocitinib, baricitinib, upadacitinib, and SHR0302).
"These findings may provide a reference for clinicians in prescribing JAK inhibitors for patients with AD," the authors write. "Further evidence from real-world data on longer-term safety [is] warranted."
Abstract/Full Text (subscription or payment may be required)