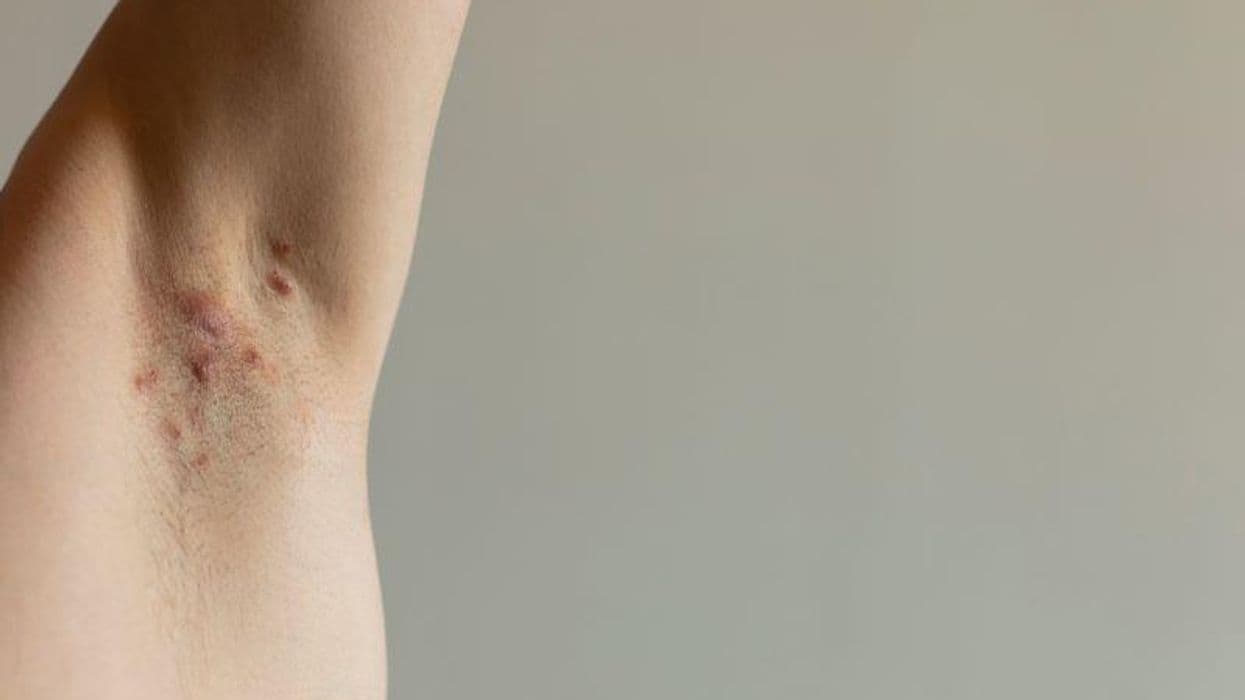
For patients with moderate-to-severe hidradenitis suppurativa (HS), bimekizumab shows promise in phase 3 trials, according to research presented at the annual meeting of the American Academy of Dermatology, held from March 17 to 21 in New Orleans.
Alexa B. Kimball, M.D., M.P.H., from Harvard Medical School in Boston, and colleagues conducted two phase 3 studies involving patients with HS (BE HEARD I with 505 participants and BE HEARD II with 509 participants). The trials included a 16-week initial and 32-week maintenance treatment period. Patients with moderate-to-severe HS were randomly assigned to receive (initial/maintenance) bimekizumab 320 mg every two weeks (Q2W)/Q2W, bimekizumab Q2W/Q4W, bimekizumab Q4W/Q4W, and placebo/bimekizumab Q2W in a 2:2:2:1 ratio. Bimekizumab Q2W/Q2W and bimekizumab Q2W/Q4W were combined to bimekizumab Q2W until week 16. The primary end point was HS 50 percent Clinical Response (HiSCR50) at week 16.
The researchers found that more patients receiving bimekizumab than placebo achieved HiSCR50 (BE HEARD I: 47.8 or 45.3 versus 28.7 percent for bimekizumab Q2W or bimekizumab Q4W versus placebo; BE HEARD II: 52.0 or 53.8 versus 32.2 percent, respectively). A significantly higher proportion of patients achieved HiSCR75 with bimekizumab versus placebo at week 16 with both dosing regimens in BE HEARD II and with Q2W in BE HEARD I. Across regimens and both studies, responses were maintained to week 48 with bimekizumab for HiSCR50 and HiSCR75. The safety profile of bimekizumab was consistent with previous studies across BE HEARD I and BE HEARD II.
"Treating moderate-to-severe cases with bimekizumab has shown promising results in Phase 3 patient trials, with sustained improvement after one year," Kimball said in a statement
The study was funded by UCB, the manufacturer of bimekizumab.