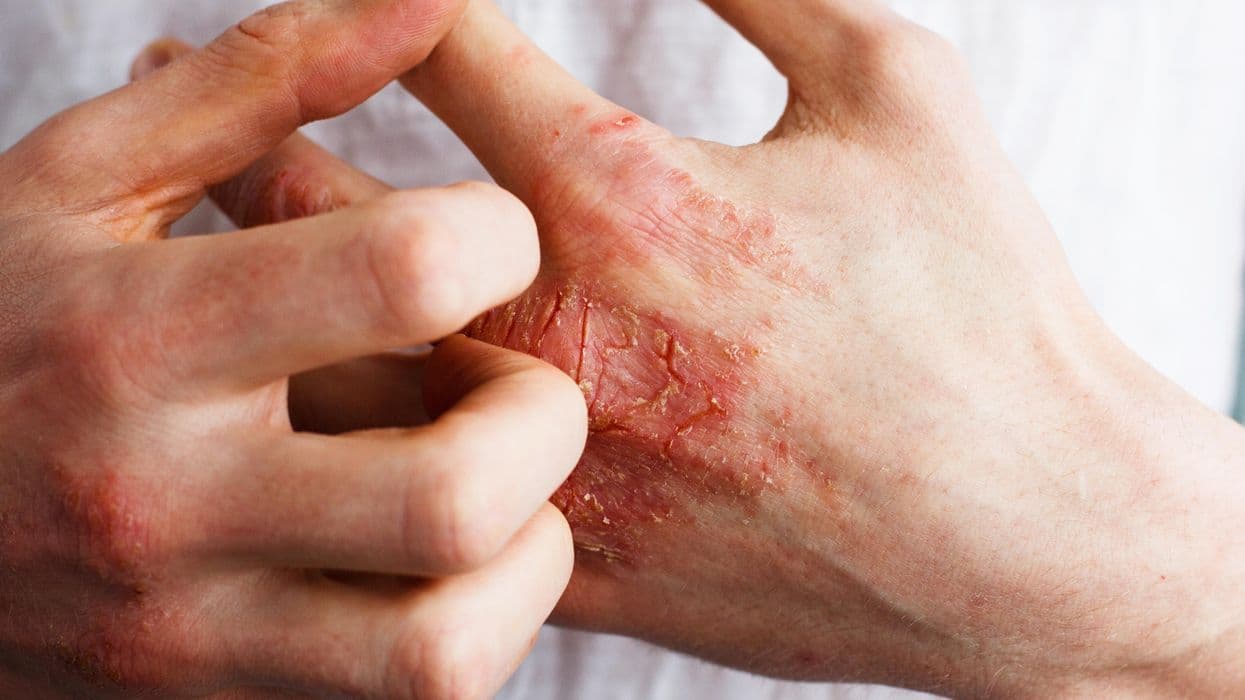
Serum Dupilumab Levels Not Linked to Treatment Response in Eczema
By Dermsquared Editorial Team | November 02, 2022
For patients with atopic dermatitis (AD) receiving dupilumab, serum dupilumab levels at 16 weeks of treatment are not associated with treatment response, according to a study published online Nov. 2 in J AMA Dermatology .
Lotte S. Spekhorst, M.D., from the University Medical Center Utrecht in the Netherlands, and colleagues examined serum dupilumab levels at 16 weeks of treatment and evaluated the association of these levels with treatment response at 52 weeks in patients with AD in a clinical, prospective, observational study. Data were included for 295 patients with AD.
The researchers found that the median drug level was 86.6 µg/mL (interquartile range, 64.6 to 110.0 µg/mL) at 16 weeks of treatment. Serum dupilumab levels did not differ significantly based on responder status at week 16. Nonsignificant odds ratios were seen for serum dupilumab levels at 16 weeks in terms of prediction of long-term response or adverse events in multivariate logistic regression.
"No association with treatment response or adverse effects to dupilumab were identified in patients with AD during the first year of dupilumab treatment," the authors write.
Several authors disclosed financial ties to pharmaceutical companies, including Sanofi and Regeneron, the manufacturers of dupilumab.
Abstract/Full Text (subscription or payment may be required)